 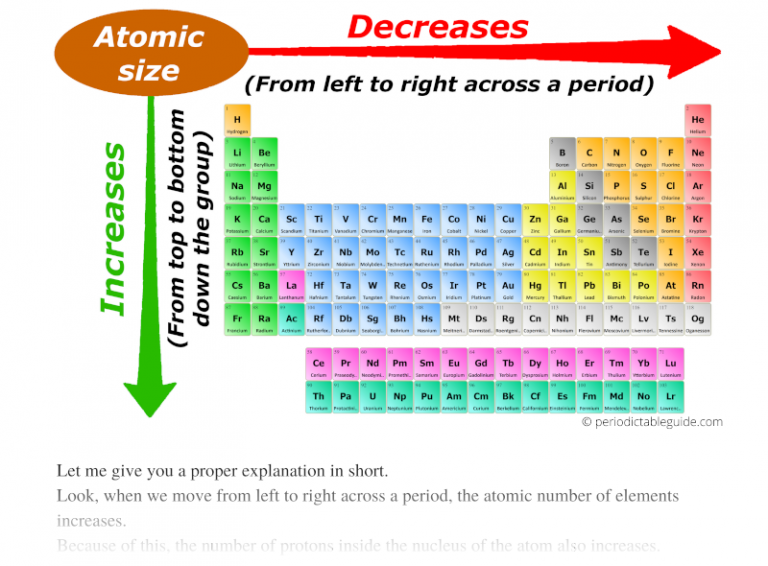
Note: This periodic trend excludes the Noble gases (Group 18). Note that the noble gases do not have an electronegativity. This is observed through Periodic Trends, as the further right of a period, and higher up a group are observed to have increased values in electronegativity. This scale ranges from 0.7 to 4.0, where cesium is the least electronegative element, and with fluorine being the most electronegative element. 
It was created by measuring the bond energy of the different elements joined by covalent bonding. The scale that was formed in order to measure this property is the Pauling scale. Which properties can be identified using periodic trends? Electronegativity:Įlectronegativity is a property that measures the tendency of an atom to attract electrons to form a bond. We discuss trends for properties such as electronegativity, atomic radius, first ionization energy, reactivity and electron affinity. This is due to the structural similarities’ elements have within a period or family that allows for these trends to take place. These trends have allowed scientists in the past to predict certain characteristics of unknown elements. For a more detailed examination of the difference, check out metals, metalloids and nonmetals.Periodic trends are observable patterns in the properties of an element that are dependent on its position in the Periodic Table. Moving the opposite way, the elements take on properties of metals. 
As you move up and across from the bottom left of the table, the elements begin to take on the characteristics of nonmetals. Metallic and Nonmetallic Characteristics are a group of physical properties. 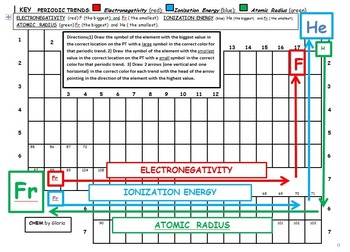
ionization energy decreases as you move down the table.ionization energy increases as you move left to right across the table.The tighter the nucleus holds an electron, the more energy needed to remove it. Ionization Energy is the energy needed to remove an electron from a gaseous atom. Atoms with low electronegativity are highly electropositive. Think of it as the opposite of electronegativity. electronegativity decreases as you move down the tableĪ related term is electropositive.electronegativity increases as you move left to right across the table.In general, the higher the electronegativity, the stronger the force of attraction between bonded electron and the atom nucleus. Both of these reduce the force of attraction between the nucleus and the added electron.Įlectronegativity is the measure of attraction between the atom’s nucleus and electrons in a chemical bond. As you move down, the outer electron is both shielded from the nucleus by filled shells and physically further away. electron affinity decreases as you move down (with exceptions)Ītoms with stronger nuclear charge tend to have higher electron affinities as you move across the table.electron affinity increases as you move left to right.It is measured by the energy change in the atom as an electron is added to the gaseous form of the atom. The outer electrons are not held as tightly as the outer electrons of the elements above them on the table.Įlectron Affinity is the ability of an atom to accept an electron. These outer shells are shielded from the positive charge of the nucleus by layers of electrons and the effective charge they experience is less than the element above them. As you move down the periodic table, the valence of the the atom remains the same, but there are more filled electron shells between the outer electrons and the positive nucleus. The electrons form shells and are attracted strongly to the positive charge in the nucleus, pulling the shells closer to the center and effectively making the atom smaller with the addition of each proton. atomic radius increases as you move downĪs you move across the periodic table from right to left, each element contains one more electron and one more proton.atomic radius increases as you move right to left.The organization of the periodic table shows the periodic trends of six different physical properties of the elements: atomic radius, electron affinity, electronegativity, ionization energy, and metallic/nonmetallic character.Ītomic radius is half the distance between two identical atoms touching each other. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
